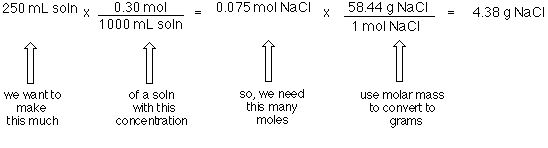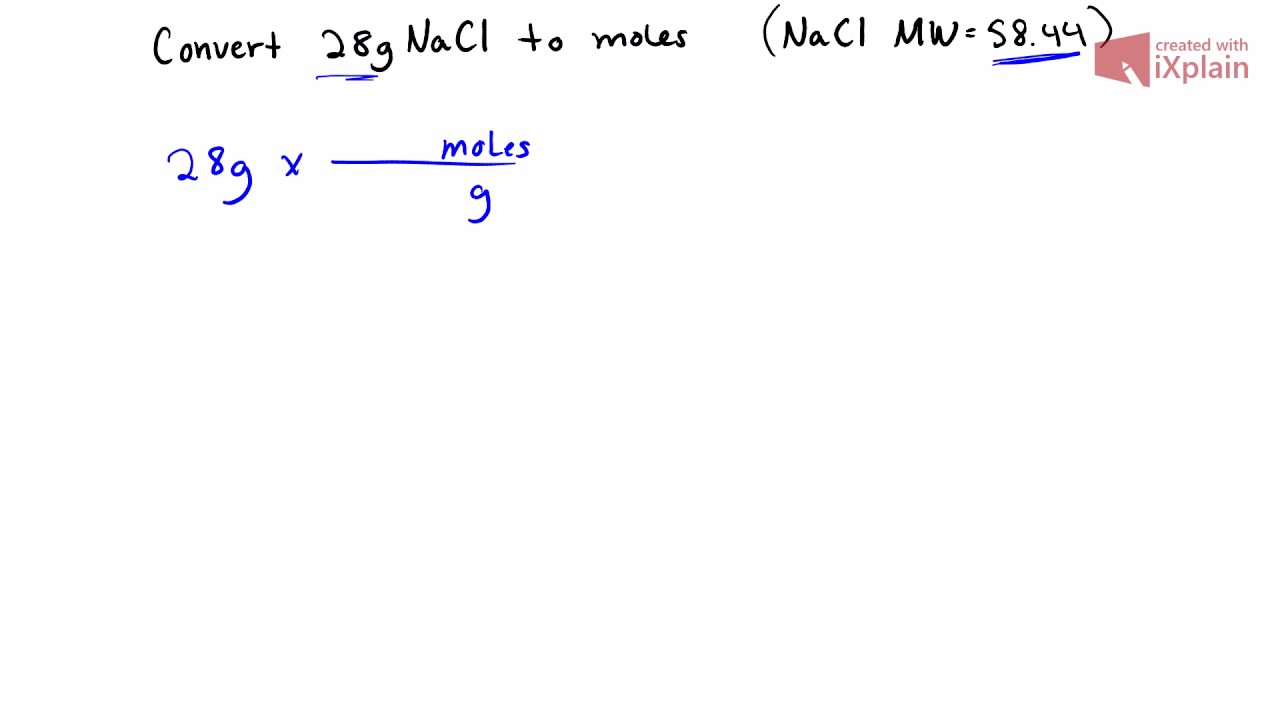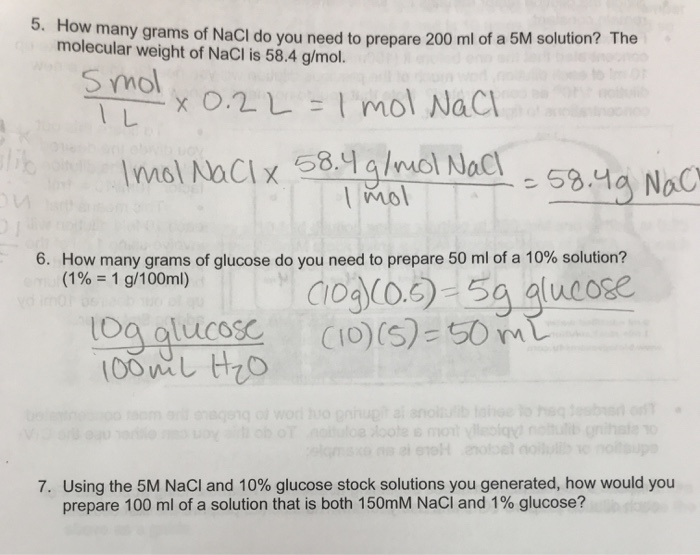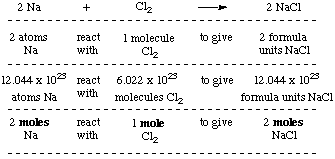The Mole & Chemical Quantities. The Mole Mole-the number of particles equal to the number of atoms in exactly 12.0 grams of carbon mol = 6.02 x. - ppt download

1. Determine the Gram Formula Mass of a compound 2. Convert between Grams and Moles 3. Convert between Moles and Grams. - ppt download

SOLVED: How many grams are in 3.40 mol of sodium chloride (NaCl)? Express your answer numerically in grams.

Calculate the mass of 1 mole of each one of the following: (a) `NaCl` , (b) `CaCO_(3)` , (c ) `FeSO - YouTube
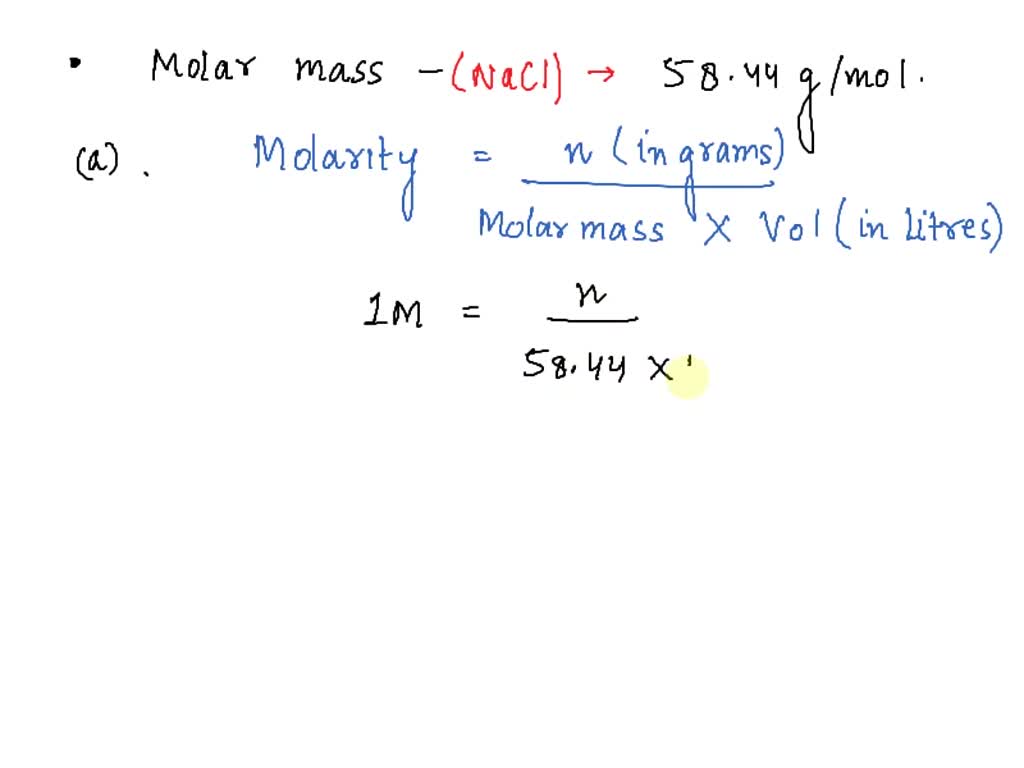
SOLVED: Molar mass of NaCl is 58.44 g/mol (this means, 1 mole of NaCl weighs 58.44 g). You are a researcher who needs to prepare several solutions. How many grams of NaCl